Lexaria Bioscience Corp’s (NASDAQ:LEXX, LEXXW) proprietary drug delivery technology, DehydraTECH, improves the way active pharmaceutical ingredients (APIs) enter the bloodstream by promoting healthier delivery methods and increasing the effectiveness of fat-soluble active molecules and drugs.
The Company’s technology can be applied to many different drugs and other active ingredients in oral formats including tablets, capsules, oral suspensions and more.

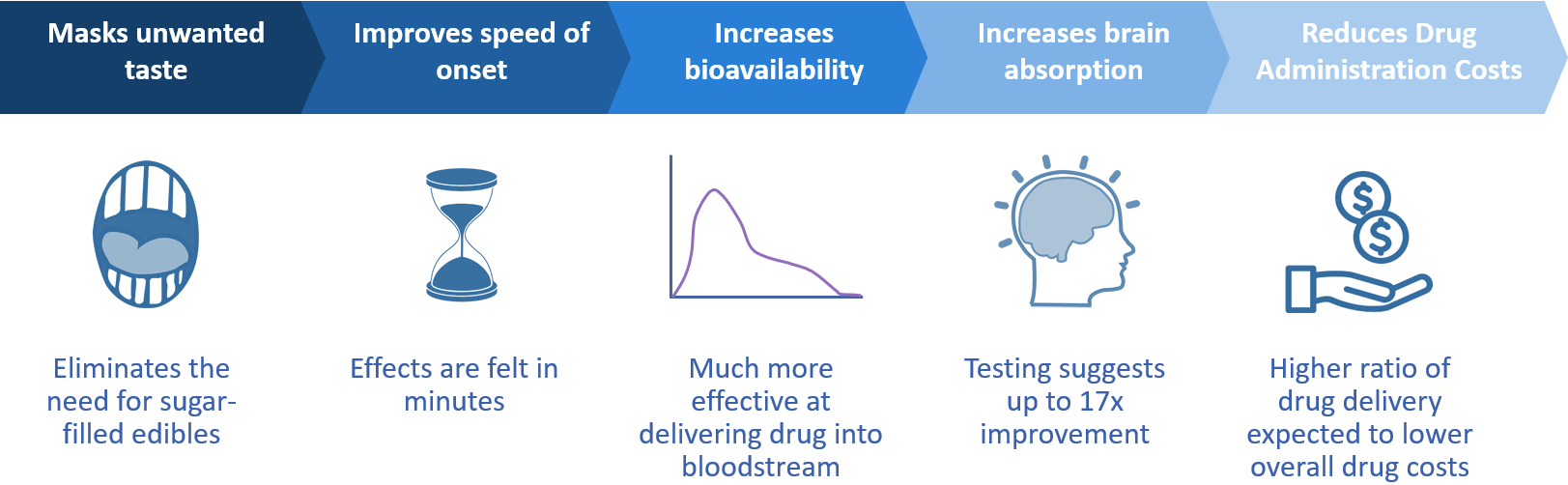
- Increases bio-absorption
- Reduces time of onset from up to 1 – 2 hours to just minutes
- Masks unwanted tastes
- Is much more effective at delivering drugs into the bloodstream
- Provides a higher ratio of drug delivery expected to lower overall drug costs
Companies in various locations around the world are examining DehydraTECH for incorporation into a broad range of products. Lexaria operates a federally licensed, in-house research laboratory and is building a robust intellectual property portfolio with 41 patents granted internationally and many more patents pending worldwide.